Suture-tape augmentation of anterior cruciate ligament reconstruction: a randomised controlled trial (STACLR) comparing residual knee laxity, patient reported outcomes and complications.
Updated on 11/06/2025 – Minh Ngo (0403781465 – Whatsapp | trucminh.ngo@wh.org.au)
Investigators: Dr Lachlan Huntington, Mr Scott Tulloch, Mr Lachlan Batty, Dr Andrew Griffith, Phong Tran | Research Personnel: Minh Ngo
All study documents are kept on SharePoint accessible only to the Investigators and Research team. Protocol available below.
Overview
The aim of this study is to compare the outcomes of ACL reconstruction using suture-tape stabilisation to ACL reconstruction without this tape. Our research looks at how stable patients’ knees are after ACL reconstruction with suture-tape stabilisation and compares this to before surgery and to their other ‘normal’ knee. This difference will be compared between the two groups (with and without suture tape), with the primary time-point being 2 years after surgery. Knee stability is measured using a testing machine called the GNRB, which is designed to measure the degree of looseness in the knee.
All participants involved in the study will have ACL reconstruction surgery performed by either Mr Scott Tulloch or Mr Lachlan Batty. Participants are to complete questionnaires about their pain, function, quality of life and return to sport status and goals as part of standard care and for study purposes. Additionally, patients are to attend surgical follow-up appointments for physical examinations at 2 weeks, 6 weeks, 3 months and 12 months post-surgery with GNRB assessments performed at 3 months, 12 months and 24 months post-surgery. The timepoints for collection of data are outlined in the figure below:
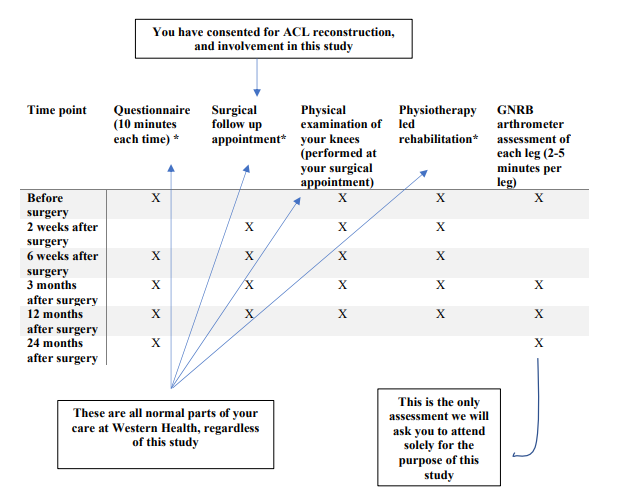
Recruitment Statistics
The goal for recruitment is 66 randomised patients. The first patient for this study was randomised on 19th May 2022 and there are currently 53 patients officially enrolled in the study since, with 17 patients having completed the study.
Here are the current recruitment and participant progress statistics for the study:
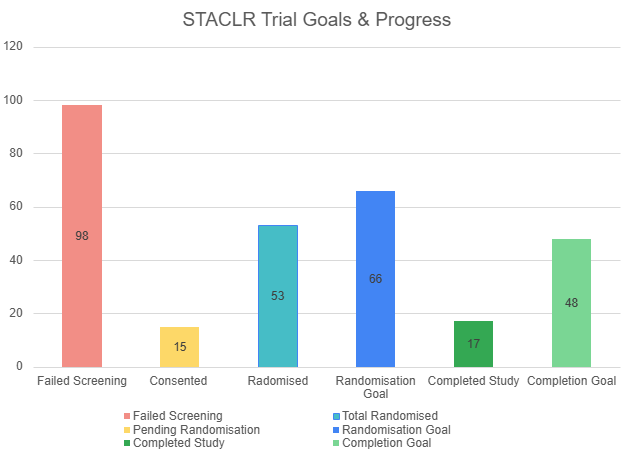
We currently have a 71% study completion rate of the primary outcome (completion of 2-year post-op GNRB assessments).
Information for Registrars
Seeing new patients through our Friday morning Consultant Clinic is generally where we will recruit suitable patients to be enrolled in this study, or follow-up patients to collect GNRB, examination and PROMs data post-operatively.
The study involves pre-operative, 3-month, 12-month and 24-month post-operative measurements of tibial translation on both the patient’s injured and uninjured knee using a GNRB machine which we have specifically obtained for this study, which lives in the Level 2 Physio Outpatient department.
If you are helping to see patients for either Scott Tulloch or Lachlan Batty in consultant clinic, please consider whether any new patients you are seeing would be appropriate for the study, and if so please get either Mr Batty or Mr Tulloch to see them to complete the clinical examination findings and then send them to Clinic D to be enrolled and consented to take part in the study and be sent for their GNRB measurements
Basically, patients are eligible for inclusion in the study if they are over 18, and do not have a concomitant injury that would alter their post-operative rehab course, such as a meniscal injury. For post-op study patients, the research staff will generally put an exam finding sheet on the patient’s clipboard to be completed – please get Mr Tulloch or Mr Batty to come and examine the patient for these findings to ensure consistency. They will still need to go to Clinic D after seeing you to get their GNRB measurements done.
Upcoming surgeries for ACL RCT patients may require additional equipment – if in doubt, check with Mr Lachlan Batty/ Mr Scott Tulloch or Andy Griffith.
If you have any questions about the day-to-day running of the trial, please contact Andy Griffith (0400875446) or Minh Truc Ngo (0403781465). For further information about the study itself, you can also contact Lachlan Huntington.
Protocol paper
Authors
Minh Ngo – 2025
