Google-translated patient education website to improve informed consent for surgery in Orthopaedic patients with limited English: a randomised controlled trial
Principal Investigator: Prof Phong Tran.
Associate Investigator: Quoc Toan Phu
Research staff: Natali Coric, Libby Spiers
Aims
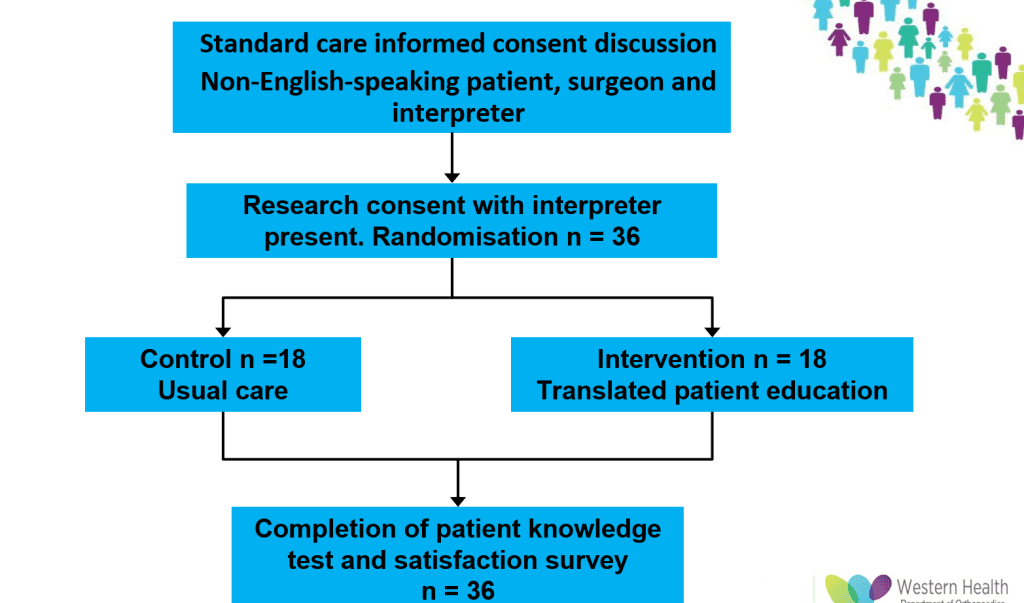
This RCT will evaluate the use of a Google-translated information website in improving informed consent for elective surgery in Orthopaedic patients with limited English. Patients who require an interpreter when providing consent for a THA or TKA will be invited to take part. They will be randomised to the usual care group (no additional intervention) or the education group, who will be provided with the WH Orthoanswer website information about their procedure translated into their preferred language using Google Translate. Both groups will complete a bespoke Knowledge Questionnaire that aims to assess patients’ knowledge of the key components that patients must understand for the consent to be legal and valid: indications, procedure, recovery, risks, benefits and alternatives. This will allow us to see whether patient knowledge is improved by reading relevant information in their preferred language.
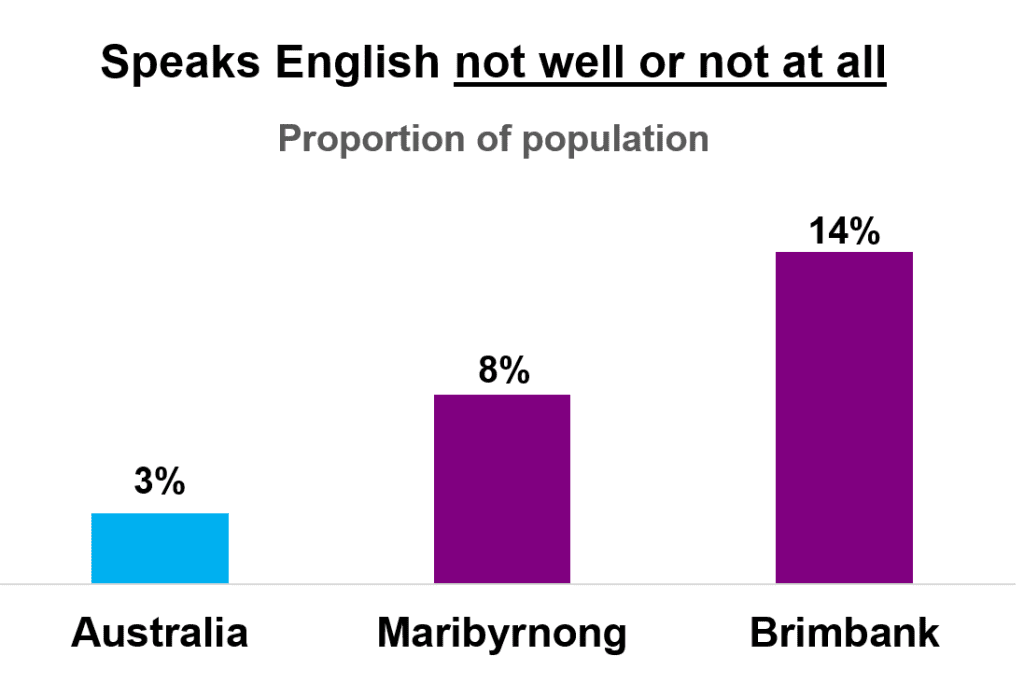
This is an important project because we know that non-English-speaking patients have poorer health outcomes and lower health literacy. Even with a professional interpreter, surgical consent is more likely to be inadequate in this group compared to patients proficient in English, with a higher risk of misunderstanding. Previous work at WH, by Fraval et al, demonstrated that in English speaking patients, supplemental education resources improve patient understanding of informed surgical consent. Dr Toan Phu has worked to get this project off the ground over the last few years, and it has been great to see that we’ve been able to get ethics approval, trial registration, and a Bank Vic research grant to support this research during 2023/4.
Inclusion criteria:
- Over 18 years
- Limited or no English proficiency, requiring an interpreter for surgical consent
- Online translation available for preferred language
- Self-reported ability to read and understand basic medical information in their preferred language
- Consented for total hip or knee arthoplasty
Exclusion criteria:
Has already used Orthoanswer website in their preferred language
Recruitment target and progress
Recruitment target is 36 participants (18 per group). The first participant was randomised on 10 Jan 2024.
Recruitment process
Background