Comparison of outcome measures and complication rates following three different approaches for primary Total Hip Arthroplasty. A pragmatic randomised controlled trial.
Update Libby Spiers 13.01.2025
All identifiable and regulatory documents kept on WH SharePoint, accessible only to Investigators. Protocol available below.
Principal Investigator: Prof Phong Tran.
Associate Investigator: Mr Adrian Talia
Research staff: Natali Coric, Libby Spiers
This RCT, initiated by Mr Adrian Talia and Prof Tran in 2017, aims to compare the outcomes of three THA surgical approaches (anterior, posterior and Hardinge) at the 2-year mark. Outcomes are measured using the Oxford Hip Score, VAS pain scale, and the EQ-5D-5L quality of life assessment. The primary outcome is the OHS at 2 years. Patients can complete their questionnaires via the online REDCap platform, over the phone or via post. This data is collected pre-operatively, and then 6 weeks, 6 months, 1- and 2-years post-op (primary time-point). Patients who consented to participate were randomly assigned to one of the approaches, rather than being allocated to a specific surgeon. Surgeons only perform the approach(es) they normally use in clinical practice.
Eligibility
Eligible participants include any adult patient who has hip osteoarthritis requiring a THR. Exclusion criteria are revision surgery, and indications for a hip replacement other than OA (i.e, fracture, malignancy, developmental dysplasia). Patients who are unable to complete their PROMs will also be excluded.
Recruitment
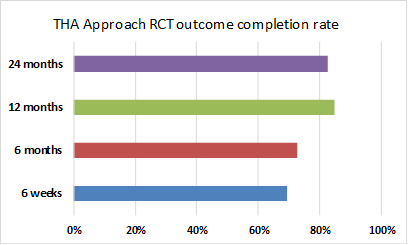
Our recruitment target is 284 participants, with 94 participants to receive each approach. Recruitment was completed in March 2024, and all surgery and 6-week data collection has also been completed.
124 patients have completed outcome measures at the primary time-point of 2 years. This is 86% of those who are still enrolled who have reached 2 years. This retention rate should allow us to reach the sample size needed at 2 years.
Study protocol paper
Authorship
2023 – Libby Spiers
